Dynamic neural states underpin motor symptom severity in Parkinson's disease: a longitudinal analysis of chronic cortico-subthalamic nucleus recordings.
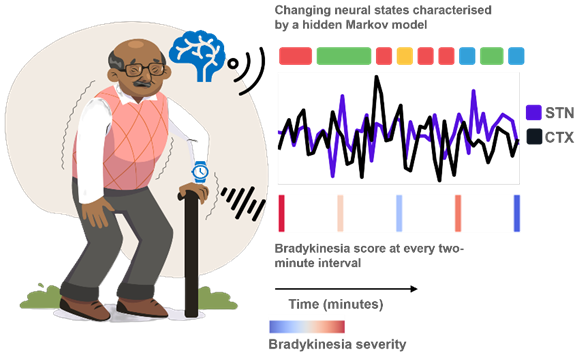
Parkinson's movement symptoms do not stem from a single abnormal brain rhythm, but from constantly shifting activity patterns across large brain networks. By analysing long-term recordings from the motor brain network, we identified specific brain "states" linked to worse slowness, tremor, or medication-induced involuntary movements, alongside others associated with better movement. This suggests that future treatments could be more effective by targeting these whole brain states rather than individual rhythms, opening the door to truly personalised brain stimulation.
Motor symptoms in Parkinson's disease (PD) may arise due to transient, network-wide neural dynamics that extend beyond beta-band oscillatory activity within the motor cortical-subthalamic nucleus (STN) circuit. We applied a four-state Hidden Markov Model (HMM) to identify states of local and interregional oscillatory synchrony from chronic motor cortical and STN recordings (1046 h from 10 hemispheres) in five patients with PD (mean age 49 years), with concurrent measurements of bradykinesia, dyskinesia and tremor quantified using wearable sensors. Neural states exhibited distinct spectral and temporal features relating to symptom severity. Two states exhibited spectral signatures-particularly STN low and high gamma, STN delta/alpha, cortical beta, and cortico-STN beta coherence-that predicted worsening bradykinesia. STN beta oscillations were not consistent predictors of bradykinesia (p = 0.52), but did predict worsening tremor (p < 0.01) and also improvements in dyskinesia severity (p < 0.001), in a state specific manner. These states also displayed compensatory features associated with bradykinesia amelioration, including cortical delta/alpha activity, cortical high gamma, and cortico-STN high gamma coherence. Additionally, we identified a state, marked by STN beta without cortico-STN beta coherence, whose increased lifetimes and occurrence improved motor function (p < 0.001). Our findings highlight the multidimensional nature of motor impairments in PD and suggest that adaptive interventions targeting state features-rather than single frequency bands-may offer new opportunities for personalised neuromodulation. AO: MRC Clinician Scientist Fellowship (MR/W024810/1), Rosetrees Trust/Race Against Dementia Team award, Oxford Hospitals Charity, and the Jon Moulton Charity Trust. TL: China Scholarship Council.
2026. Brain (e-Pub ahead of print).
2023. Mov Disord, 38(3):423-434.
2022. NPJ Parkinsons Dis, 8(1):88.
2025. Neurobiol Dis, 207:106858.